 I must preface this next post with a little truth in advertising. I’m a chemist. True blue, to my very core. College degree in physical chemistry, PhD in chemistry. So when I heard about a cable show on AMC whose whole premise rested on a chemistry teacher manufacturing meth, I must say, I was slightly skeptical. The propensity for letdown was huge, both in plot and in science. Well, let me assure you that Breaking Bad broke good. A “Break”out hit in its second season, the show has managed to layer complex serialized storytelling with compelling characters and stories, and even better science. In fact, chemistry itself can very well be considered a recurring character on this show and we’ll highlight some of the best moments in a bit.
I must preface this next post with a little truth in advertising. I’m a chemist. True blue, to my very core. College degree in physical chemistry, PhD in chemistry. So when I heard about a cable show on AMC whose whole premise rested on a chemistry teacher manufacturing meth, I must say, I was slightly skeptical. The propensity for letdown was huge, both in plot and in science. Well, let me assure you that Breaking Bad broke good. A “Break”out hit in its second season, the show has managed to layer complex serialized storytelling with compelling characters and stories, and even better science. In fact, chemistry itself can very well be considered a recurring character on this show and we’ll highlight some of the best moments in a bit.
ScriptPhD Grade: A+
The Premise
If the pilot episode doesn’t get your attention in the first five minutes, then I don’t know what will. A man wearing nothing but his skivvies and a gas mask careens a Winnebago in the New Mexico desert, a passed out body beside him, two more dead in the back, and a toxic sludge of chemicals seeping on the floor. With impending sirens approaching, he videotapes a final goodbye and apology to his family. Through flashbacks, we come to find out that the man is Walter White, an unassuming chemistry teacher in Albuquerque, NM. While on his humiliating moonlighting shift as a car wash attendant, because we pay our public school teachers so well, Walt collapses. The culprit? Lung cancer. Terminal. Inoperable. He decides to infuse some excitement into his life on a bust ride with his brother-in-law, a DEA agent. Only instead of discouraging Walt, the bust shows him how much money can be made. While pondering the possibility of leaving his family financially secure after his passing, he spots an old flunky student, Jesse Pinkman, fleeing the scene. “You know the business, I know the chemistry,” he proposes to Jesse. An idea is born, and the metamorphosis of Walter White begins. Back to the original scene, the sirens turn out to be fire trucks, one of the many hair-raising escapes to come, and Walt and Jesse live to sell meth another day.
In addition to Walt (played by the talented Bryan Cranston), and Jesse (dazzling newcomer Aaron Paul), we meet Skyler (Anna Gunn), Walt’s supportive but perplexed wife, who grows to be very suspicious of him as he has a harder time curtailing his clandestine activities, and Walt, Jr., a teenager with Cerebral Palsy, sensitively portrayed by RJ Mitte. The relationships serve as a centerpiece of the show are unraveled like the plot, in layers and tantalizingly. As Walt’s own family unit faces turmoil, Jesse, too, is disowned by his for his drug use. What started out as a business transaction between a teacher and former student blossoms into a tender father-son relationship. Meanwhile, while Walt’s well-meaning DEA brother-in-law Hank (Dean Norris) closes in on the hottest new meth dealer in town, Walt and Jesse face a series of personal and professional setbacks. For every two steps forward, for every dollar made, there is a new foe, a new nemesis, or new unintended collateral. All of the action culminates in an electrifying Season 2 finale sure to generate buzz and anticipation for Season 3.
The Science
Science on Breaking Bad is given the red carpet treatment: it’s sleek, sexy, geek-chic, tongue-in-cheek and everywhere. The show revels in delightful touches such as the title credits interspersing elements from the periodic table. Walt’s classes brim with interesting blink-or-you-miss-it factoids, such as H. Tracy Hall inventing the first reproducible process for making diamonds. To a stupefied, gun-happy Jesse, he makes the suggestion of killing a drug lord with castor beans, the source of the protein toxin ricin. And let’s not mention the two separate synthetic methods he comes up with to cook and crystallize the best meth the New Mexico DEA has ever seen. The darkly comedic highlights of the show are Walt and Jesse’s interactions in their “laboratory”, a beaten-down Winnebago camper. Shocked by Jesse’s sloppy street cooking, Walt pilfers glassware and equipment from his classroom—gas masks, round bottom flasks, reflux condensers, crystallization dishes—to build a setup worthy of Pfizer. Along the way, Jesse gets some remedial chemistry that he failed back in high school. I mean, sure, they’re making a devastating and highly illegal narcotic, but at least it’s via a proper Grignard reagent amination of a Schiff base!
On a more serious note, Breaking Bad also strives for a VERY candid and unrelenting portrayal of both cancer and the ramifications of the modern-day drug trade. Often whitewashed in entertainment, Walt’s cancer, and the side effects are shown in a brutal way, but the stark realism also underscores his desperation as the illness unfolds. Easily on par with David Simon’s brilliant The Wire on HBO, in the world of Breaking Bad no one is absolved from the intertwining effects of drugs—the rising body count, both from use and dealing, the strain on law enforcement, and families torn apart. In an astute opening TRULY ripped from the headlines, a Season 2 Breaking Bad episode starts with an original narcocorrido, a Mexican drug ballad evolved from its folk music tradition that is often used to chronicle the drug trade and escalating violence over the last two decades. Take a look:
Bottom line: the science is white-hot, the writing is red-hot, the meth is blue and the humor is black, so why aren’t you watching?
Accolades
Breaking Bad has been the recipient of a number of recent awards and critical acclaim. They won a 2009 Peabody Award for excellence in television achievement. Bryan Cranston won the 2008 Emmy for Outstanding Leading Actor in a Dramatic Series. Series creator and executive producer Vince Gilligan won a Writers Guild of America award for the Pilot episode. Many more achievements are sure to come for their outstanding sophomore effort!
For the ScriptPhD.com Top 4 Walter White Chemistry Moments in the show thus far and an in-depth discussion of the neat science behind them, click “continue reading”…
Top 4 Walter White Chemistry Moments
4) A salt and battery…
Sometimes, desperate situations call for desperate (and clever!) chemical measures. The sticky wicket? Jesse, as you will come to learn when you watch this show, is not the sharpest knife in the drawer. While on an exotic synthetic staycation in the lovely New Mexico desert, he stores the key to his Winnebago-cum-meth lab in the ignition. Two days later, the result is a dead battery and no one around for miles to help. D’oh! To make matters worse, their spare generator catches on fire, they run out of cell phone charge, and the only person who knows how to come get them gets lost. Walt suggests rebuilding a battery.
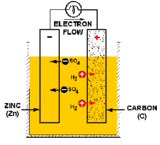
A battery (or a voltaic cell) is really just a series of fuel cells that store combined chemical energy to provide a high source of voltage power. And a fuel cell is an electrochemical device, or a galvanic cell (named after its inventor, Luigi Galvani), which converts free energy of a chemical reaction into electrical energy, or electricity. Generate electricity, generate charge, find a way to collect said charge, you’ve got power. Simple, right? In the case of batteries, the basic building block is a primary cell (also called a simple galvanic cell), which is made of four components: an anode electrode (the negative end), a cathode electrode (the positive end), an electrolyte solution that will generate positive and negatively charged ions for the two chemical reactions that take place, and a conductor (usually a wire) to carry the current of the electrons from one side to the other. The flow of charge ALWAYS goes from the cathode (the positive end) to the anode (the negative end).

Now that we are armed with the basics, here’s how Walt built his galvanic cells. For the anode (or source of negative charge) he uses a galvanized metal, specifically zinc, which they get by collecting coins and spare metallic parts (nuts, bolts, washers, etc). For the cathode (the positive charge where the current will flow out), Walt uses graphite and mercuric oxide that he ground down from the Winnebago’s brake pads. For the electrolyte, he soaks a sponge in potassium hydroxide (KOH). So in this case, referring back to the picture of the galvanic cell, the flow of positive charge will come in the form of K+ ions, and the negative charge from the OH– ions. Remember, we also need a conductor to carry the current of electrons—the charge—from one half-cell to the other. Walt uses copper wire, which he then connects to the jumper cables by collecting all six cells and pooling the electric current to restart the van’s battery.
Realistically speaking would this actually restart the camper? Sadly, probably not. We’ve talked about what a galvanic cell is, now let’s talk about how it works. The charge, or electric current, is generated by two separate chemical reactions (or half-reactions) that occur on either electrode of the cell. At the anode, an oxidization reaction strips electrons from the electrode (usually a metal of some sort), resulting in overall negative charge. At the cathode, free electrons that have traveled through the conducting circuit are used to reduce the electrode species to generate a positive charge. Taken together, these two values add up to the total cell potential, defined as the ability to force electrons through a circuit, and measured in voltage.
The zinc-carbon cell that Walt has built is a variation of a classic Bunsen cell, and we can estimate the cell voltage at around 2 total Volts. If we break down the two half-reactions, oxidation and reduction, we can add up the total potential using a fancy-schmancy mathematical equation called the Nernst equation. The potential of the mercuric oxide reduction, used in commercially available mercuric oxide batteries, is 1.35 Volts, and the potential of zinc oxidization is -0.76 Volts. To determine the full potential of a cell, we subtract the anode from the cathode, and this reaction adds up to 2.1 Volts. Walt mentions that he only has material enough for six cells. So if we add this up, we get enough voltage, give or take, to build a typical 12 Volt battery. Perfect in theory. But, when we talk about real-life circuits, we have to factor in electrical resistance, the opposition of electric current. Resistance occurs in all sorts of conductors, including metals (due to electron scattering) and ionic liquids (depending on concentration and insularity of the solution medium). If we’re going to get super-technical, we could invoke a physics property called Ohm’s Law which states that the ultimate current (which we measure in amperes) potential is inversely proportional to its resistance. The higher the resistance, the lower the ultimate current. So while Walt was able to build a battery with the proper voltage, the internal resistance would likely not generate enough current to provide the immense power necessary to jump-start a car that big. Most cars use a standard 12 Volt battery, but for cold-cracking need about 400-600 Amps, higher if you are going to start something like the Winnebago. The battery that Walt built would probably generate 20-30 Amps of current, based on current for a typical basic Galvanic cell, and that’s being generous. But how many shows on the air are even attempting this kind of clever science? So Breaking Bad gets an A for effort and that’s just all there is to it!
Incidentally, you can easily make your own galvanic cell at home. Check out this neat video of six lemons powering a low-wattage LED bulb:
Lemon Battery – Watch more funny videos here
3) What’s your name?
Of course, every drug kingpin worth his weight in meth has to have a nom de guerre. Mr. White’s sobriquet choice? Heisenberg. I must admit my inner chemistry geek did major cartwheels when I heard this. It’s such an appropriate name for him on so many levels. Heisenberg, of course, is a tongue-in-cheek reference to Werner Heisenberg, one of the great physicists of our time. He is the father of the “Heisenberg Uncertainty Principle” in quantum mechanics, which states that measured values for a particle’s position and momentum cannot be ascertained simultaneously with equal precision. It turns out that particles actually act more like waves, whose three-dimensional spatial function can be separated into three axes, or directions, x, y and z, horizontal, vertical and diagonal, respectively. So, if for a certain particle we can specify momentum with absolute certainty, then its position can be found anywhere along those axes with an equal probability. For this contribution, Werner won a Nobel Prize in physics in 1932.
What a clever and ironic name for Walt, whose own life and identity faces so much uncertainty as the show unfolds: in his cancer diagnosis, his long-term prognosis, his deteriorating relationship with his family, his precarious one with Jesse, and the theoretical uncertainty of his motivations for manufacturing meth, which certainly evolve over the two first seasons.
It’s important to note Heisenberg is also a very controversial historical figure. He played a prominent role in the German nuclear energy project, the race to develop an atomic bomb prior to World War II, although never formally affiliated with the Nazi movement. He later joined a prominent group of scientists who opposed the use of tactical nuclear weapons as warfare. The Tony-award winning play “Copenhagen” centered around the possibilities of a mysterious meeting in 1941 between Heisenberg and fellow physicist Neils Bohr in German-occupied Copenhagen around which there remains to this day… well… uncertainty.
A big ScriptPhD props to the writing team on Breaking Bad for this little gem!
2) It’s enough to just melt you!
Not since the CSI Season 2 episode “Bully For You”, in which a victim’s body found in a duffel bag decomposed so badly it had to be poured out, has a liquefied body played such an important role on a television episode. You’ve just killed the drug dealer who was supposed to peddle your batch of crystal meth, but you have to make the body disappear quietly without your DEA brother-in-law finding out. I mean seriously, folks, we’ve all been there, right? “The best thing to do,” Walt concludes, “is dissolving [the body] in strong acid.” We’ll talk more about this in a second, but first, check out the minisode for the episode “The Cat’s in the Bag…”. Pay particular attention to the disastrous results of this suggestion in the last 1:30, especially if you have a strong stomach.
In the episode, Walt asks Jesse to pick up a plastic container in which to dissolve the body. Even Jesse, dubious of this, says to him, “Any decent acid is gonna eat right through this.” “Not hydrofluoric [acid],” Walt concludes. What? An acid that can eat through flesh and bones but will leave a flimsy plastic bucket intact? This sounds like crazy talk! Well, actually, he’s right. An acid, chemically speaking, is nothing more than a compound that is able to give up a proton to a willing base in solution. By proton we mean a positively-charged hydrogen (H+) with water acting as the accepting base in most cases. The stronger the acid, the more readily it gives up this proton, and the higher the concentration of H3O+ protonated water ions floating in solution, called the dissociated state. Why wouldn’t it react with plastic? Walt tells Jesse to look at the bottom of the bins for a symbol called “LDPE”. He’s talking about low-density polyethylene, the plastic polymer that makes up everything from plastic bags, various containers, dispensing bottles, wash bottles, tubing, to molded laboratory equipment. The repeating units of CH2, some linked by side branches, make for a strong, compact bond and very low reactivity. That is, this chemical will neither give up its hydrogens when exposed to a solution, nor will it accept any. In this case, the acid will primarily disintegrate the body, which as we know is about 80% water, thus creating a solution, but the nonreactive hydrocarbon of the bin would remain chemically inert.
Hydrofluoric acid, contrary to Walt’s statement, is NOT really that strong of an acid, compared to other options, but has some interesting properties useful for Walt and Jesse’s predicament. Back up for a moment. There’s two ultimate determinants of acid strength—electronegativity (the charge on the molecule that the hydrogen is attached to) and the size of the atom that the hydrogen is attached to, which has to do with the strength of the bond. As you move across the periodic table, atom charge strongly increases, which is why fluorine is a much better acid candidate than nitrogen or an unreactive atom like carbon. BUT, as you move down the periodic table, elements get bigger and bigger, and as their size increases the strength of the bond with hydrogen weakens for various reasons. (We’ll skip discussion of covalent bond orbitals for next time!) So acidically speaking, HF < HCl (stomach acid) < HBr < HI. Personally, I really would have chosen a very concentrated hydrochloric acid or sulfuric acid solution like John George “The Acid Murderer” Haigh, or better yet, liquid lye (the base NaOH), which would be able to react with the organic components of tissue and fat content and wouldn’t have dissolved Jesse’s tub, but where’s the drama in that? Realistically speaking, HF would not be able to liquefy the body to the substantial extent shown in the episode. But because it is a weak acid and exists primarily in its undissociated state, it is able to penetrate deeply into the skin before deprtotonating, thus making it an excellent and efficient corrosive for human flesh (and very dangerous!). That’s right, it eats your body from the inside out. More importantly, it reacts strongly with calcium and magnesium, so it would be able to efficiently dissolve Emilio’s bones for disposal. [Incidentally, when people are treated at the hospital for HF poisoning, the first line of treatment is calcium gluconate to “compete” with the calcium in your bones as a neutralization reaction.] Unfortunately, it also reacts with silicon dioxide, the major component of glass and ceramics. Hence the look of absolute and profound horror on Walt’s face as Liquid Emilio, the bathtub, and everything in-between come crashing down.
Take home lesson? If you’re desperate to get rid of a dead body by chemical disincorporation, for God’s sakes, read up on your chemistry first!
Annnnnnd ::drumroll:: the best Walter White chemistry moment to date?
1) A little tweak of chemistry!


Without question, the scientific highlight of the show to me, thus far, is the integration of clever reaction chemistry to advance a major plot on the show. Early in the episode “Crazy Handful of Nothin’”, Mr. White is teaching his class about the power of chemical reactivity. Sometimes, it’s gradual and imperceptible, to which he gives the example of metal oxidation. But other times, the reaction is violent, quick, and produces tons of energy. On the chalkboard, he writes an example of an explosive compound, mercury fulminate, Hg(ONC)2. Relatively easy to make synthetically, fulminated mercury is a powerful explosive and was long used as a detonation primer for dynamite.
This chemistry lesson proves to be a dynamite primer of its own later in the episode. The first good large batch of meth—1 pound!—that he and Jesse were able to synthesize was stolen without compensation by Albuquerque’s toughest drug wholesaler. What’s a wronged high school chemistry teacher to do? Walt shows up at Tuco’s casa with a large crystal of what looks like more meth and a demand for his money. But Walt surprises him by announcing that it isn’t in fact meth, and throws the crystal to the ground. The result? KABOOM! See the before and after pictures. What is that stuff, a stunned Tuco asks. “A little tweak of chemistry.” Most notable about this scene is that it caps the season-long transformation of Walt’s character. As he walks over to his car, money in hand, reputation restored, he hasn’t just earned the respect of a thug. This is the moment in the show that delineated a before and an after, where the line was drawn in the sand, and we never again saw the innocent sick chemistry teacher desperate to save his family. A drug dealer was born.

The only slight criticism that I have is that in reality, mercury fulminate wouldn’t actually look like the large meth-like crystals portrayed on the show. The chemical synthesis process usually leads to very fine white powder precipitate crystals like the one in the picture on the right. But we can just assume that Walt used his past career as a “Crystallographer Extraordinaire” to produce the largest mercury fulminate crystals to date ;-). Secondly, this stuff is unstable and extremely reactive. As in don’t touch it, don’t expose it to light, don’t mess with it unstable. It can be detonated by sparks, shock, friction, or even a wayward glance. Realistically speaking, Tuco handling the stuff with his knife and dropping it on the table would be enough to ignite it. We’ll also have to assume Walt was very careful in handling an entire plastic bag of it. Nevertheless, this whole scene is so creative, well-written and downright badass, that here at ScriptPhD.com, we can’t allow minor quibbles to detract from the moment.
The season finale of Breaking Bad aired May 31, 2009 on AMC, but for those of you not yet watching this spectacular series, there is plenty of time to catch up. The Season 1 DVD is available in stores and the Season 2 DVD release date will be updated on our site as soon as we know it! And we’re going to do our darndest to welcome Vince Gilligan, the show’s brilliant creator and executive producer, to our in-house ScriptPhD lab to chat all things Breaking Bad! Stay tuned!
For a look ahead at Season 3, check out this sneak peek preview:
All video clips and pictures are © 2007-2009 AMC Television and Sony Pictures Television. All rights reserved.
~*ScriptPhD*~
Wow, looks great! I wanna start watching it now. =)
Just a thought, perhaps you should hook up with Amazon.com to get commissions off the DVD saless of these shows/movies. 😉
LOL! We do what we do for the love, not the money ;-). Mostly we’ll be looking to interview talent and writers and make people aware of what the very best of tv and film is.
But that’s a very good idea!
I have never heard of this show – but damn it looks awesome. And you know, with the whole teaching aspect of it, I’m even more drawn in. Science is a really hard subject to teach to kids – especially with really hard concepts to grasp, so although I wouldn’t teach my kids to make meth, it’d be interesting to see what else he teaches us in the episodes that i could use in the classroom.
I really like your post. Does it copyright protected?
Hi, gr8 post thanks for posting. Information is useful!
How soon will you update your blog? I’m interested in reading some more information on this issue.
Hi Gary. There have been a number of posts since this one. Please feel free to check out the rest of the blog. Cheers and thanks for the support!
Hi! I like your srticle and I would like very much to read some more information on this issue. Will you post some more?
I actually thought that scene was Walt “stepping up”… not quite there yet. But stepping up to the call of duty. To me, Walt started being a true drug dealer at the end of one of the episodes this season. He at first tried to help out a fellow meth-maker at the store, but then seeing him talking to his partner in the parking lot, walks right up to him and says: “Stay out of my territory”.
the phosphorus in the water or whatever in the first season supposedly making phosgene is false too. cocl2 doesn’t even have a phosphorus in the compound.
About the fuel cells (point 4).
As I was watching the episode, I sensed that such a device would never ever supply power enough to start a Diesel engine; in the meantime, I was wondering why Walt was not usisg a way smaller amount of so-generated electricity to recharge his phone! Even easier, he could switch his SIM card with the out-of-range, but still working Jesses’s mobile. Silly, isn’t it?
Guido that is a REALLY good point! I forgot about Jesse’s phone. It was definitely a clever concept that was not married by realistic chemistry.
When Walt says that the “supply of current will flow out from” the positive end is he correct? My understanding is that the current flows from negative to positive. I believe that’s what the diagram above says too. Is this a mistake, a misinterpretation on my part, or is he right?
Mercury fulminate is not as sensitive as described here. Yes sparks, and flames will set it off. Scooping it with a knife or pouring it would more than likely not set it off. A hammer would set it off. If it were so unstable, it would never have found a use it the explosives industry. Unlike like nitrogen triiodide, which has no use and is so unstable you can’t move it. Also, one wouldn’t ever attempt to crystallize mercury fulminate as internal strains in the crystal will make it much more sensitive and impossible to handle.
Steve K and Chemist — You’ve both caught some pretty excellent flaws in the chemistry stories on Breaking Bad.
What particularly bugged me was exactly what Chemist points out, that Walt likely would not have been able to even handle the fulminated mercury to begin with, let alone that the violence of the explosion was exactly exaggerated.
Steve K — It’s been quite a while since I’ve seen the episode where they built the battery. I’d have to rewatch to see if he did indeed reverse the order of the electron flow.
I took HS chemistry a while ago, but I remember a little bit. But I don’t understand why methylamine cannot be synthesized from methanol and ammonia. Can you explain it please?
He definitely got the flow mixed up when he sad the current flows from positive to negative, however he could be using the common convention employed when drawing circuit diagrams. Since Benjamin Franklin initially guessed incorrectly we have been stuck with it and practically it doesn’t really matter so I won’t fault him for it.
You have anode (positive) and cathode (negative) mixed up in your description above.
I believe it was mixed up on the episode of the show. I’ll have to rewatch it to see if the error was on our end. If so, we will correct it. Thanks!
I can confirm, having watched the episode in question an hour ago, that they mixed it up on the show. Walt definitely describes the cathode as the positive side of his cell, which is wrong. So, from A to F in one fell swoop! Let’s hope nobody has followed those instructions and received a nasty shock!
Top Walter White moments
6. Dissolved body in hydrofluoric acid
5. Made raison poison out of castor beans
4. Used water and shaved match striker strips to make deadly phosphine gas
3. Made battery out of galvanized coins,nuts & brake pads & potassium hydroxide
2. Wiped hard drive inside evidence room using a 252V electromagnet inside car
1. Used fulminated mercury (looks like meth, got through security) as explosive
They actually DIDN’T get anode and cathode mixed up. The anode is not positive in a discharging battery, only in a recharging one.
Yay to Gwynn and Chemist! Anode and cathode refer to what the electrode is doing, not their polarity- removing and donating electrons to the inside of the device, respectively. Mercury fulminate is relatively stable, perhaps Admin had it confused with silver fulminate, which is much more shock and scratch sensitive.